Printable Copy of Chapter Summeries
| regents_review.doc | |
| File Size: | 261 kb |
| File Type: | doc |
Atomic Concepts
1. Dalton – basic unit of matter is the atom; cannon ball model
2. JJ Thomson – used cathode ray tube; found electrons; “plum pudding model”
3. Rutherford – gold foil experiment; small, dense, positively charged nucleus; mostly empty spac
4. Bohr – nucleus surrounded by electrons in circular orbits
5. Atoms have a positive charged nucleus, surrounded by negatively charged electrons
6. Protons and neutrons are found in the nucleus
7. Protons are positive, neutrons have no charge, electrons are negative
8. In an atom, # protons = # electrons (no charge = neutral atom)
9. Mass of a proton and neutron are 1 amu; electron < 1 amu
10. Orbitals – regions of the most probable electron location
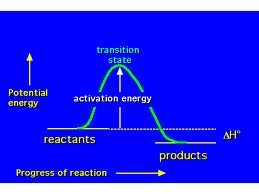
11. Each electron in an atom has its own specific amount of energy
12. Electrons have a higher energy state in the excited state
13. Energy is released when electrons fall back (excited à ground)
14. Bright light spectrum is produced when electrons return to ground state
15. Valence electrons – outermost electrons
16. Valence electrons affect chemical properties of an element
17. Isotopes – same number of protons, different number of neutrons
18. Atomic mass – average of all naturally occurring isotopes
19. # neutrons = mass # - atomic #
20. Positive ion = lost electrons; negative ion = gained electrons
21. Ground state electron configuration matches periodic table; excited state does not
22. Lewis dot structure; a dot represents a valence electron (# dots = # valence electrons)
23. *** be able to calculate the atomic mass of an element, given masses and ratios of isotopes (% ÷ 100 * mass and add them all together)
Nuclear Chemistry
1. Stability of an isotope is based on the ratio of neutrons to protons in the nucleus
2. Most nuclei are stable. Some are unstable –they will spontaneously decay, emitting radiation
3. Each radioactive isotope has a specific model and rate of decay. (Table N + O)
4. Transmutations – a change in the nucleus of an atom that converts it from one element to another. This can occur naturally or can be artificial by the bombardment of the nucleus by high energy particles ( + β à)
5. Spontaneous decay – release of alpha, beta, positrons and/or gamma radiation from the nucleus of an unstable isotope
6. Use table O for mass, charge, symbol (gamma has the most penetrating power b/c small mass and no charge)
7. Fusion: Hydrogen + Hydrogen à Helium + energy (only on the sun)
8. Fission – is the splitting of a heavy nucleus into 2 lighter nuclei. U-235, Pu- 239
9. Fusion – requires very high temperature
10. Fission – produces radioactive wastes that remain for long periods of time
11. Radon-222, Krypton-85, N-16 are radioactive wastes that can be safely dispersed into the environment
12. Solve nuclear equations by making the mass and charge = on both sides of the à
13. Energy released in a nuclear reaction (fission or fusion) comes from a fractional amount of mass converted into energy. Nuclear changes convert matter into energy
14. Energy released during nuclear reactions is much greater than energy released during chemical reactions
15. Risks associated with radioactivity and the use of radioactive isotopes include: biological exposure (causing cancer), long term storage, disposal, and nuclear
accidents
16. Radioactive isotopes have many beneficial uses:
Medicine: short half life, quick elimination from body
Tc-99 brain tumor
I-131 thyroid disorder
Ra and Co-60 for cancer treatment
Age: Living things – C-14 : C-12
Of rocks, minerals – U-238: Pb-206
17. Half life problems:
# half life periods= total time
Time of one half-life (table N)
Formulas/Math of Chemistry
1. Compounds – can be chemically decomposed
2. A compound is a substance composed of 2 or more elements chemically combined
3. Empirical formula – simplest whole number ratio of atoms
4. Molecular formula – the actual ratio of the atoms in a molecule
5. Structural formula – the arrangement of atoms shown
6. There is a conservation of mass, energy and charge in all chemical reactions
7. Balance equations: # atoms reactants = # atoms products (add coefficients)
8. Formula mass – sum of the atomic masses of the atoms
9. Gram formula mass (molar mass) – one mole of a given substance
10. %
= part * 100 Note: Part = mass of element asked about
whole Whole = gram formula mass
11. % hydrate = part * 100
whole
12. Types of chemical reactions:
Synthesis: two substances combine to make one product; A + B à AB
Decomposition: compound is broken down into simpler substances: AB à A + B
Single Replacement: Element + Compound à “New” element + “New” compound A + BC àB
Double Replacement: Compound + Compound à“New” compound + “New” compound AB + CD àAD + CB
13. Naming:
One oxidation state (charge): metal + nonmetal -ide
MgCl2
magnesium chloride
One oxidation state (charge) w/ Table E: Never change name of table E ion
Na2SO4
sodium sulfate
More than one oxidation state (charge): Roman Numerals
metal (Roman Numeral) + nonmetal –ide
Fe2O3
iron (III) oxide
Nonmetals: Use prefixes
P2O5
diphosphorous pentoxide
14. Formula Writing:
1.) Criss-cross oxidation #’s (charges)
2.) reduce
Calcium Nitrate Magnesium Sulfate copper (II) chloride
Ca+2(NO3)-1 Mg+2(SO4)-2 Cu+2Cl-1
Ca(NO3)2 MgSO4 CuCl2
Periodic Table
1. Elements are arranged according to atomic number
2. An elements location gives you an indication of physical and chemical properties
3. Atomic # = # of protons; identifies element
4. Mass # = # protons + # neutrons
5. Metals- left of staircase; metalloids- on staircase; non-metals- right of staircase
6. Noble gases- group 18
7. Metals- good conductors of heat and electricity, malleable and ductile
8. Elements can be differentiated by physical properties and by chemical properties
9. Groups 1,2,13-18- elements within the group have the same # valence electrons (except He)
10. Elements within the same group have similar properties
11. Use Reference table S to predict trends (across a period, down a group)
12. Non-metals-poor conductors of heat and electricity, brittle solids, gases
Bonding
1. Compounds can be differentiated by their chemical and physical properties
2. Ionic compounds (metal-nonmetal); poor conductors as solids- good as liquids, aqueous
3. Molecular compounds (nonmetal-nonmetal); covalent bonding
4. Ionic bond- transfer of electrons; metal-nonmetal
5. Polar covalent bond- unequal sharing of electrons; nonmetal-nonmetal
6. Non polar covalent bond- equal sharing of electrons; diatomics (HOFBrINCl’s)
7. Metallic Bonding- “sea of mobile electrons;” metals only
8. Triple bond- three pairs of electrons shared; double bond- two pairs shared
9. Shape and distribution of charge decides molecular polarity
10. Nonpolar molecules- symmetrical charge; CO2, CH4, CCl4, diatomics
11. Polar molecules- asymmetrical charge; HCl, NH3 and H2O
12. Negative ions- gains electron(s), radius increases
13. Positive ions- loses electron(s), radius decreases
14. When a bond is broken, energy is absorbed
15. When a bond is formed, energy is released
16. Atoms bond together to obtain a total of 8 valence electrons (become stable)
17. Noble gases- stable valence configurations; tend to not bond
18. Hydrogen bonding- H and FON; strong; high melting/boiling points; low vapor pressure
19. Dipole forces- (polar molecules) weak; low melting points, high vapor pressure; soluble in water, conduct as aqueous
20. Nonpolar- weak; insoluble in water, low melting points, high vapor pressure, conduct as aqueous
21. Lewis dot- one dot = one valence electron (**for compounds you must get formula first)
22. Electronegativity- a measurement of an atoms ability to gain electrons
23. Most polar or most ionic- largest electronegativity difference
24. Least polar or least ionic- smallest electronegativity difference
25. Both ionic and covalent bonds- polyatomic ions- hint answer must have at least 3 elements
Physical Behavior of Matter
1. Matter is classified as a pure substance (element or compound) or as a mixture
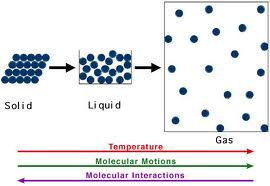
2. Phases of matter- solid (definite shape and volume); liquid (definite volume, shape of container); gas (no definite shape or volume- takes shape and volume of
container)
3. Particle Models:
1. Dalton – basic unit of matter is the atom; cannon ball model
2. JJ Thomson – used cathode ray tube; found electrons; “plum pudding model”
3. Rutherford – gold foil experiment; small, dense, positively charged nucleus; mostly empty spac
4. Bohr – nucleus surrounded by electrons in circular orbits
5. Atoms have a positive charged nucleus, surrounded by negatively charged electrons
6. Protons and neutrons are found in the nucleus
7. Protons are positive, neutrons have no charge, electrons are negative
8. In an atom, # protons = # electrons (no charge = neutral atom)
9. Mass of a proton and neutron are 1 amu; electron < 1 amu
10. Orbitals – regions of the most probable electron location
11. Each electron in an atom has its own specific amount of energy
12. Electrons have a higher energy state in the excited state
13. Energy is released when electrons fall back (excited à ground)
14. Bright light spectrum is produced when electrons return to ground state
15. Valence electrons – outermost electrons
16. Valence electrons affect chemical properties of an element
17. Isotopes – same number of protons, different number of neutrons
18. Atomic mass – average of all naturally occurring isotopes
19. # neutrons = mass # - atomic #
20. Positive ion = lost electrons; negative ion = gained electrons
21. Ground state electron configuration matches periodic table; excited state does not
22. Lewis dot structure; a dot represents a valence electron (# dots = # valence electrons)
23. *** be able to calculate the atomic mass of an element, given masses and ratios of isotopes (% ÷ 100 * mass and add them all together)
Nuclear Chemistry
1. Stability of an isotope is based on the ratio of neutrons to protons in the nucleus
2. Most nuclei are stable. Some are unstable –they will spontaneously decay, emitting radiation
3. Each radioactive isotope has a specific model and rate of decay. (Table N + O)
4. Transmutations – a change in the nucleus of an atom that converts it from one element to another. This can occur naturally or can be artificial by the bombardment of the nucleus by high energy particles ( + β à)
5. Spontaneous decay – release of alpha, beta, positrons and/or gamma radiation from the nucleus of an unstable isotope
6. Use table O for mass, charge, symbol (gamma has the most penetrating power b/c small mass and no charge)
7. Fusion: Hydrogen + Hydrogen à Helium + energy (only on the sun)
8. Fission – is the splitting of a heavy nucleus into 2 lighter nuclei. U-235, Pu- 239
9. Fusion – requires very high temperature
10. Fission – produces radioactive wastes that remain for long periods of time
11. Radon-222, Krypton-85, N-16 are radioactive wastes that can be safely dispersed into the environment
12. Solve nuclear equations by making the mass and charge = on both sides of the à
13. Energy released in a nuclear reaction (fission or fusion) comes from a fractional amount of mass converted into energy. Nuclear changes convert matter into energy
14. Energy released during nuclear reactions is much greater than energy released during chemical reactions
15. Risks associated with radioactivity and the use of radioactive isotopes include: biological exposure (causing cancer), long term storage, disposal, and nuclear
accidents
16. Radioactive isotopes have many beneficial uses:
Medicine: short half life, quick elimination from body
Tc-99 brain tumor
I-131 thyroid disorder
Ra and Co-60 for cancer treatment
Age: Living things – C-14 : C-12
Of rocks, minerals – U-238: Pb-206
17. Half life problems:
# half life periods= total time
Time of one half-life (table N)
Formulas/Math of Chemistry
1. Compounds – can be chemically decomposed
2. A compound is a substance composed of 2 or more elements chemically combined
3. Empirical formula – simplest whole number ratio of atoms
4. Molecular formula – the actual ratio of the atoms in a molecule
5. Structural formula – the arrangement of atoms shown
6. There is a conservation of mass, energy and charge in all chemical reactions
7. Balance equations: # atoms reactants = # atoms products (add coefficients)
8. Formula mass – sum of the atomic masses of the atoms
9. Gram formula mass (molar mass) – one mole of a given substance
10. %
= part * 100 Note: Part = mass of element asked about
whole Whole = gram formula mass
11. % hydrate = part * 100
whole
12. Types of chemical reactions:
Synthesis: two substances combine to make one product; A + B à AB
Decomposition: compound is broken down into simpler substances: AB à A + B
Single Replacement: Element + Compound à “New” element + “New” compound A + BC àB
Double Replacement: Compound + Compound à“New” compound + “New” compound AB + CD àAD + CB
13. Naming:
One oxidation state (charge): metal + nonmetal -ide
MgCl2
magnesium chloride
One oxidation state (charge) w/ Table E: Never change name of table E ion
Na2SO4
sodium sulfate
More than one oxidation state (charge): Roman Numerals
metal (Roman Numeral) + nonmetal –ide
Fe2O3
iron (III) oxide
Nonmetals: Use prefixes
P2O5
diphosphorous pentoxide
14. Formula Writing:
1.) Criss-cross oxidation #’s (charges)
2.) reduce
Calcium Nitrate Magnesium Sulfate copper (II) chloride
Ca+2(NO3)-1 Mg+2(SO4)-2 Cu+2Cl-1
Ca(NO3)2 MgSO4 CuCl2
Periodic Table
1. Elements are arranged according to atomic number
2. An elements location gives you an indication of physical and chemical properties
3. Atomic # = # of protons; identifies element
4. Mass # = # protons + # neutrons
5. Metals- left of staircase; metalloids- on staircase; non-metals- right of staircase
6. Noble gases- group 18
7. Metals- good conductors of heat and electricity, malleable and ductile
8. Elements can be differentiated by physical properties and by chemical properties
9. Groups 1,2,13-18- elements within the group have the same # valence electrons (except He)
10. Elements within the same group have similar properties
11. Use Reference table S to predict trends (across a period, down a group)
12. Non-metals-poor conductors of heat and electricity, brittle solids, gases
Bonding
1. Compounds can be differentiated by their chemical and physical properties
2. Ionic compounds (metal-nonmetal); poor conductors as solids- good as liquids, aqueous
3. Molecular compounds (nonmetal-nonmetal); covalent bonding
4. Ionic bond- transfer of electrons; metal-nonmetal
5. Polar covalent bond- unequal sharing of electrons; nonmetal-nonmetal
6. Non polar covalent bond- equal sharing of electrons; diatomics (HOFBrINCl’s)
7. Metallic Bonding- “sea of mobile electrons;” metals only
8. Triple bond- three pairs of electrons shared; double bond- two pairs shared
9. Shape and distribution of charge decides molecular polarity
10. Nonpolar molecules- symmetrical charge; CO2, CH4, CCl4, diatomics
11. Polar molecules- asymmetrical charge; HCl, NH3 and H2O
12. Negative ions- gains electron(s), radius increases
13. Positive ions- loses electron(s), radius decreases
14. When a bond is broken, energy is absorbed
15. When a bond is formed, energy is released
16. Atoms bond together to obtain a total of 8 valence electrons (become stable)
17. Noble gases- stable valence configurations; tend to not bond
18. Hydrogen bonding- H and FON; strong; high melting/boiling points; low vapor pressure
19. Dipole forces- (polar molecules) weak; low melting points, high vapor pressure; soluble in water, conduct as aqueous
20. Nonpolar- weak; insoluble in water, low melting points, high vapor pressure, conduct as aqueous
21. Lewis dot- one dot = one valence electron (**for compounds you must get formula first)
22. Electronegativity- a measurement of an atoms ability to gain electrons
23. Most polar or most ionic- largest electronegativity difference
24. Least polar or least ionic- smallest electronegativity difference
25. Both ionic and covalent bonds- polyatomic ions- hint answer must have at least 3 elements
Physical Behavior of Matter
1. Matter is classified as a pure substance (element or compound) or as a mixture
2. Phases of matter- solid (definite shape and volume); liquid (definite volume, shape of container); gas (no definite shape or volume- takes shape and volume of
container)
3. Particle Models:
4. A pure substance (element or compound)- alike properties throughout (homogeneous)
5. Elements- atoms with the same atomic number; cannot be chemically decomposed
6. Mixtures- two or more substances that can be separated by physical change
7. Homogeneous mixture- (aq) uniform throughout; Heterogeneous mixture- sand, soil, garbage, not alike throughout
8. Each component of a mixture maintains its original properties
9. You can separate the components of a mixture by its
properties: density, particle size, molecular polarity, boiling point (distillation), solubility (filtration), vapor pressure (chromatography)
10. Solution-homogeneous mixture of a solute dissolved in a solvent. Solubility depends on temperature, pressure and chemical nature of the
solute and solvent
11. Molarity- concentration of a solution, use formula on table T (M = mol/L)
12. Adding solute causes the boiling point to rise and freezing point to lower
13. Parts per million (ppm)- grams solute/grams solution x 1,000,000 (**** be sure to add together solute and solvent mass for “grams of solution”)
14. Forms of energy- chemical, electrical, electromagnetic, thermal, mechanical, nuclear
15. Heat is transferred from a body of high temperature to low temperature
16. Temperature-not a form of energy; measurement of the average kinetic energy
17. Ideal gas- explains the behavior of gases; conditions; low pressure, high temperature; He and H behave most like ideal gases
18. KMT states- all gas particles are in constant random motion; volume is negligible, no forces of attraction; collisions result in a transfer of energy (total
remains constant)
19. Collision theory- reaction will occur with proper energy and orientation
20. Equal volumes of a gas have equal numbers of particles (molecules)
21. Physical change- rearrangement of existing particles in a substance
22. Chemical change- results in the formation of different substances with changed properties look for the word react
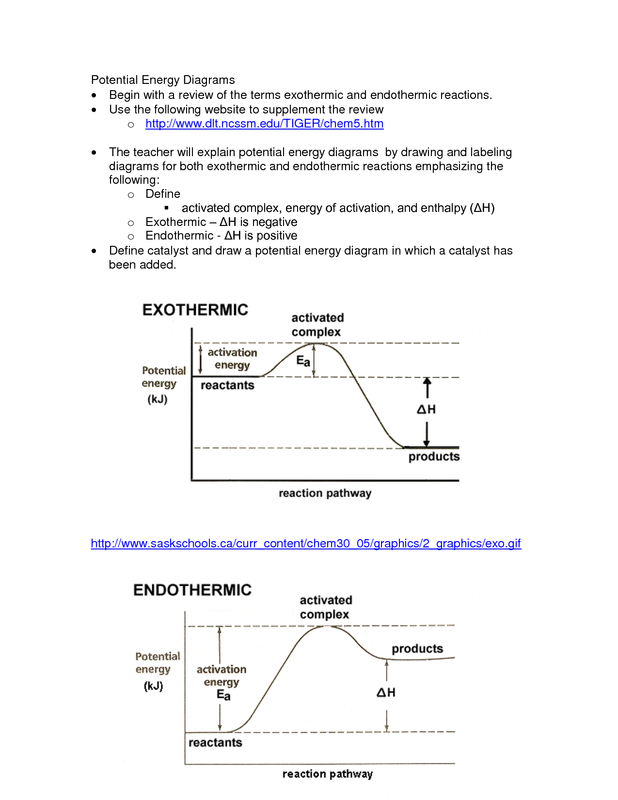
23. Chemical and physical changes can be endothermic or exothermic
24. Structure and arrangement of particles- determines the physical state of a substance at a given temperature and pressure
25. Hydrogen bonding- strong intermolecular force
26. Temperature and pressure directly related, temperature and volume directly related; pressure and volume indirectly related
27. P1V1 = P2V2 Table T – Temp in K
T1 T2
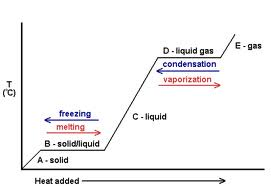
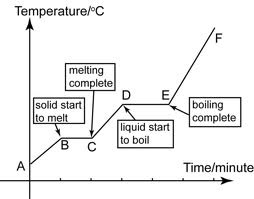
28. Heat of fusion- amount of heat needed to change a solid to a liquid at constant temp
29. Heat of vaporization- amount of heat needed to change a liquid to a gas at constant temp
30. Sublimation-solid to gas directly no liquid; weak intermolecular forces, high vapor pressure
31. Deposition-gas directly to solid
32. On heating/cooling curve- Kinetic energy increases/decreases during temperature change, remains the same during phase changes; Potential energy is opposite of KE
33. Heating/Cooling Curve:
5. Elements- atoms with the same atomic number; cannot be chemically decomposed
6. Mixtures- two or more substances that can be separated by physical change
7. Homogeneous mixture- (aq) uniform throughout; Heterogeneous mixture- sand, soil, garbage, not alike throughout
8. Each component of a mixture maintains its original properties
9. You can separate the components of a mixture by its
properties: density, particle size, molecular polarity, boiling point (distillation), solubility (filtration), vapor pressure (chromatography)
10. Solution-homogeneous mixture of a solute dissolved in a solvent. Solubility depends on temperature, pressure and chemical nature of the
solute and solvent
11. Molarity- concentration of a solution, use formula on table T (M = mol/L)
12. Adding solute causes the boiling point to rise and freezing point to lower
13. Parts per million (ppm)- grams solute/grams solution x 1,000,000 (**** be sure to add together solute and solvent mass for “grams of solution”)
14. Forms of energy- chemical, electrical, electromagnetic, thermal, mechanical, nuclear
15. Heat is transferred from a body of high temperature to low temperature
16. Temperature-not a form of energy; measurement of the average kinetic energy
17. Ideal gas- explains the behavior of gases; conditions; low pressure, high temperature; He and H behave most like ideal gases
18. KMT states- all gas particles are in constant random motion; volume is negligible, no forces of attraction; collisions result in a transfer of energy (total
remains constant)
19. Collision theory- reaction will occur with proper energy and orientation
20. Equal volumes of a gas have equal numbers of particles (molecules)
21. Physical change- rearrangement of existing particles in a substance
22. Chemical change- results in the formation of different substances with changed properties look for the word react
23. Chemical and physical changes can be endothermic or exothermic
24. Structure and arrangement of particles- determines the physical state of a substance at a given temperature and pressure
25. Hydrogen bonding- strong intermolecular force
26. Temperature and pressure directly related, temperature and volume directly related; pressure and volume indirectly related
27. P1V1 = P2V2 Table T – Temp in K
T1 T2
28. Heat of fusion- amount of heat needed to change a solid to a liquid at constant temp
29. Heat of vaporization- amount of heat needed to change a liquid to a gas at constant temp
30. Sublimation-solid to gas directly no liquid; weak intermolecular forces, high vapor pressure
31. Deposition-gas directly to solid
32. On heating/cooling curve- Kinetic energy increases/decreases during temperature change, remains the same during phase changes; Potential energy is opposite of KE
33. Heating/Cooling Curve:
Equilibrium
1. Equilibrium-rates of opposing reactions are equal; Concentration of reactants and products remain constant
2. Some chemical and physical changes can reach equilibrium
3. Entropy is a measure of the randomness or disorder of a system. A system with greater disorder has greater entropy
S à L à G
increase in entropy
G à L à S
decrease in entropy
4. Systems in nature tend to undergo changes toward lower energy and higher entropy
5. Le Chatelier’s principle can be used to predict the effect of stress (change in pressure, volume, concentration, and temperature) on a system at equilibrium
Stress Rule Hint
Increase Temp Favor endothermic Follows arrow away from energy
Decrease Temp Favor exothermic Follows arrow towards energy
Increase Concentration Favors reaction that will deplete concentration Follows arrow away from conc.
Decrease Concentration Favors reaction that will replenish concentration Follows arrow towards conc.
Increase Pressure Follows arrow towards lower gas coefficient
Decrease Pressure Follows arrow towards higher gas coefficient
HINT:
Increase: Add-Away
Decrease: Take-Toward
Kinetics
1. Collision theory- a reaction is most likely to occur if reactant particles collide with the proper energy and orientation
and the presence of a catalyst.
2. Rate of reaction depends on-temperature, concentration, nature of reactants, surface area, and the presence of a catalystWhy? Increase the frequency and effectiveness of collisions
1. Equilibrium-rates of opposing reactions are equal; Concentration of reactants and products remain constant
2. Some chemical and physical changes can reach equilibrium
3. Entropy is a measure of the randomness or disorder of a system. A system with greater disorder has greater entropy
S à L à G
increase in entropy
G à L à S
decrease in entropy
4. Systems in nature tend to undergo changes toward lower energy and higher entropy
5. Le Chatelier’s principle can be used to predict the effect of stress (change in pressure, volume, concentration, and temperature) on a system at equilibrium
Stress Rule Hint
Increase Temp Favor endothermic Follows arrow away from energy
Decrease Temp Favor exothermic Follows arrow towards energy
Increase Concentration Favors reaction that will deplete concentration Follows arrow away from conc.
Decrease Concentration Favors reaction that will replenish concentration Follows arrow towards conc.
Increase Pressure Follows arrow towards lower gas coefficient
Decrease Pressure Follows arrow towards higher gas coefficient
HINT:
Increase: Add-Away
Decrease: Take-Toward
Kinetics
1. Collision theory- a reaction is most likely to occur if reactant particles collide with the proper energy and orientation
and the presence of a catalyst.
2. Rate of reaction depends on-temperature, concentration, nature of reactants, surface area, and the presence of a catalystWhy? Increase the frequency and effectiveness of collisions
Acids, Bases, and Salts
1. Arrhenius theory- describes the behavior of many acids and bases
2. Arrhenius acids and bases are electrolytes
3. Electrolyte-is a substance when dissolved in water (aq), forms a solution capable of conducting an electrical current
4. More ions = better conductor
5. Arrhenius acids- yield H+, hydrogen ions as the only positive ion in solution
6. H+, the hydrogen ion may also be written as H3O+, the hydronium ion
7. Arrhenius bases yield OH-, the hydroxide ion as the only negative ion in solution
8. Table K and L list common bases and acids in order of strength
9. Acid + Base à Salt + H2O = acid-base neutralization Ex. HCl + NaOH à NaCl + H2O
10. Titration- laboratory process in which a volume of solution of known concentration is used to determine the concentration of another solution. Buret-glassware used for titration lab Formula-MAVA = MBVB -Table T
11. Acid-proton donor; Base-proton acceptor (hint: count H’s- in a pair, the one with more H’s is the acid, less is the base)
13. Indicators-
Acid-Litmus paper is red; Phenolphthalein is clear
Base-Litmus paper is blue; Phenolphthalein is pink
*****Use table M- Indicators
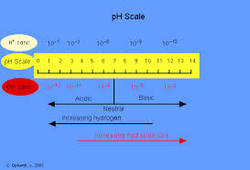
14. On the pH scale, each decrease of one unit of pH represents a tenfold increase in the hydronium ion concentration.
Ex. pH = 2, [H3O+] = 0.01 M
pH = 1, [H3O+] = 0.1 M à
concentration increases by 10 with a change from 1 to 2 in pH scale
Organic Chemistry
1. Organic compounds contain carbon
2. Organic compounds- nonpolar; react slower; nonelectrolytes (except organic acids); low melting point
3. Carbon can bond to one another with a single, double or triple bond (must have 4 bonds)
4. Table P-# of carbons with prefix
5. Hydrocarbon-compound that contain carbon and hydrogen only
6. Saturated hydrocarbon- (table Q) alkanes; CnH2n+2; all single bonds
7. Unsaturated hydrocarbons- (table Q) alkenes; CnH2n; one double bond alkynes; CnH2n-2; one triple bond
8. Use table R to identify, draw and name organic acids, alcohols, esters, aldehydes, ethers, halides, amines, amides, amino acids
9. Functional groups impart distinctive physical and chemical properties to organic compounds
10. Isomers-same molecular formula but different structural formulas (different properties)
11. Multiple covalent bonds- more than one pair of electrons shared
12. Substitution- Alkane à 2 Products
13. Addition- Alkene or Alkyne à 1 Product
14. Fermentation- Glucose à Ethanol + CO2
15. Esterification- Organic Acid + Alcohol à Ester + Water
16. Saponification- Making of soap from a fat (look for Na)
17. Combustion- Hydrocarbon + O2 à CO2 + H2O + heat
18. Polymer-long chain of repeating units (monomers)
19. Natural polymer- proteins, starch, cellulose
20. Synthetic polymers- polyXXXXX, nylon, rubber, plastics
21. Condensation polymerization- linking of monomers by the removal of water
22. Addition polymerization- linking of monomers by the “opening” of double bonds
Oxidation-Reduction
1. Oxidation-reduction (redox) reaction- involves a transfer of electrons
2. Reduction- gain of electrons
3. Half reaction- for reduction: + e- à (e- on the reactants side)
4. Oxidation- loss of electrons
5. Half reaction- for oxidation: à + e- (e- on the products side)
6. In a redox reaction the # of electrons lost = # of electrons gained
7. Assigning oxidation numbers: sum of the oxidation #’s = 0
O2-except: H2O2, OF2, Na2O2 H+1, H-1 with group 1,2 elements
8. Changes in oxidation numbers indicates that oxidation and reduction has occurred
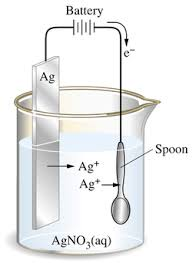
9. An electrochemical cell can either be voltaic or electrolytic
10. In an electrochemical cell: oxidation occurs at the anode, reduction occurs at the cathode
11. A voltaic cell spontaneously converts chemical energy to electrical energy
12. An electrolytic cell requires electrical energy (a battery) to produce chemical change. This process is known as electrolysis.
13. Table J (activity series):